GILUPI
Key Facts
| Invested since | 2006 |
| Based in | Potsdam |
About the company
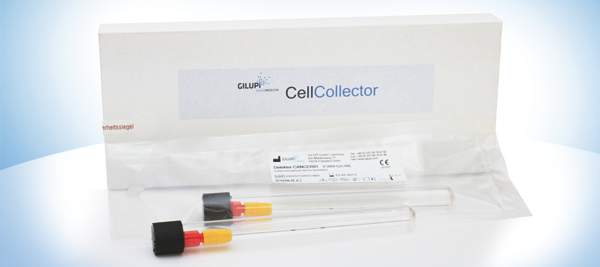
GILUPI GmbH is a medical device company with focus on the development and production of innovative products for the in vivo isolation of rare cells from the circulation. Currently, the main focus of GILUPI is the diagnostics market for cancer.
Individual oncological targeted therapies will become more and more important in tomorrow’s personalized medicine. The identification of the right drug for the specific patient is the upcoming challenge. To address this medical need, the application of the GILUPI CellCollector™ enriches rare cells (circulating tumor cells- CTCs) by directly “fishing” them in the patient’s bloodstream. By using special diagnostic analyses, these isolated cells can be characterized and/or analyzed at a molecular level. Thus, statements regarding the current status of the disease or/and the prognosis can be made. In addition, the physician is able to adjust the treatment immediately and optimize the therapy of the patient.
The GILUPI CellCollector™ is the first in vivo CTC isolation product worldwide with CE approval.
Do you want to
know more about this company?
GILUPI in the news
 zum Artikel
zum Artikel
The GILUPI GmbH is expanding to the Asian markets and received the CE-certificate for its CellCollector
 zum Artikel
zum Artikel